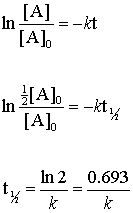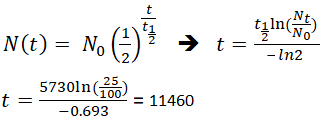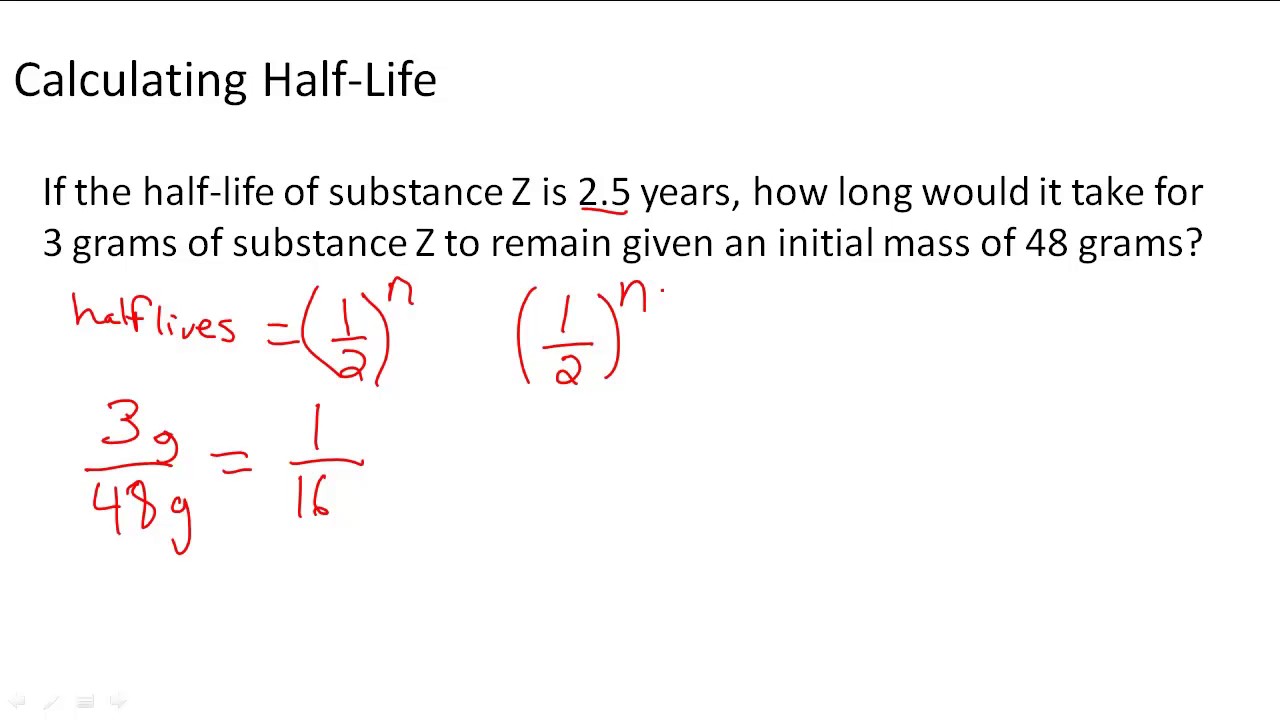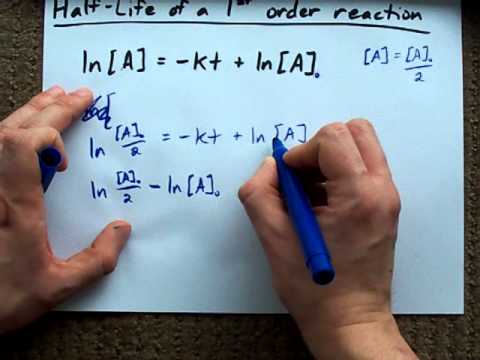half life formula chemistry
The half-life of a reaction is the time required for the reactant concentration to decrease to one-half its initial value. The half-life for the radioactive decay of 14C is 5730 years.
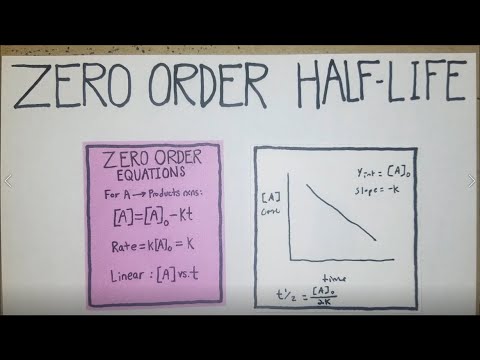
How To Calculate Half Life For Zero Order Reactions Youtube
Given that the half-life of Pd is 4 days calculate the initial mass of the sample.
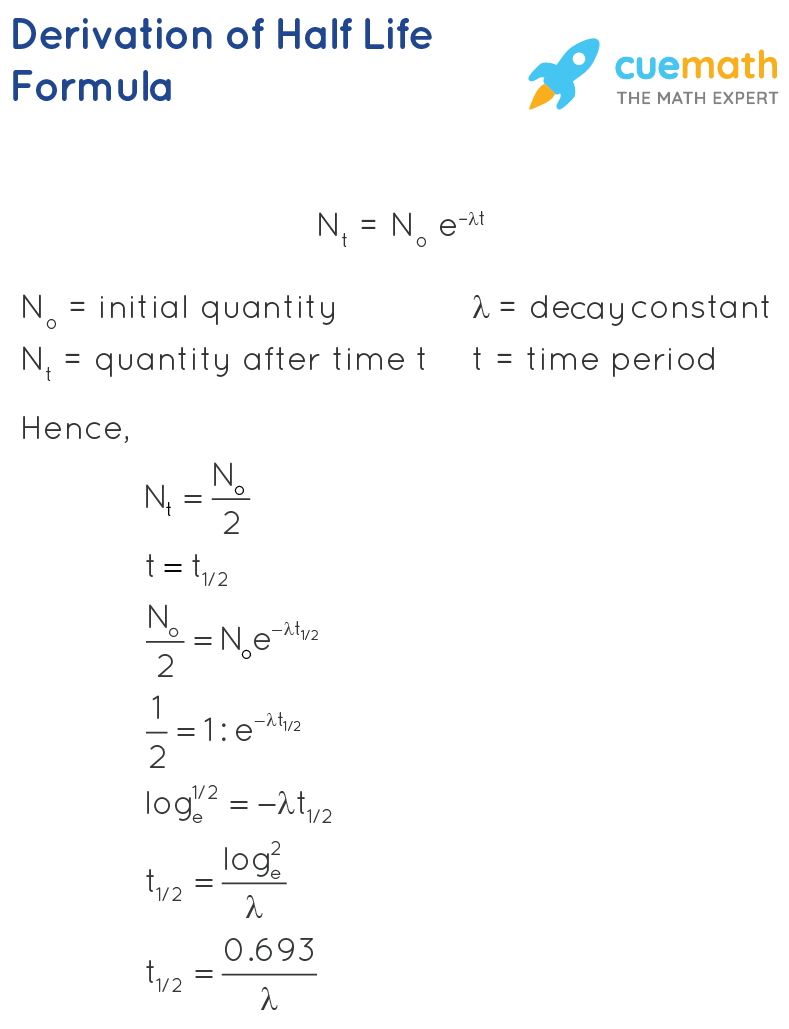
. The formula for a half-life is T12 ln2 λ. In order to calculate the half-life of a chemical specie integrated half-life equations are used according to the order of reactions. Half-Life is the required time for a sample of radioactive material to reach half the mass of the original sample.
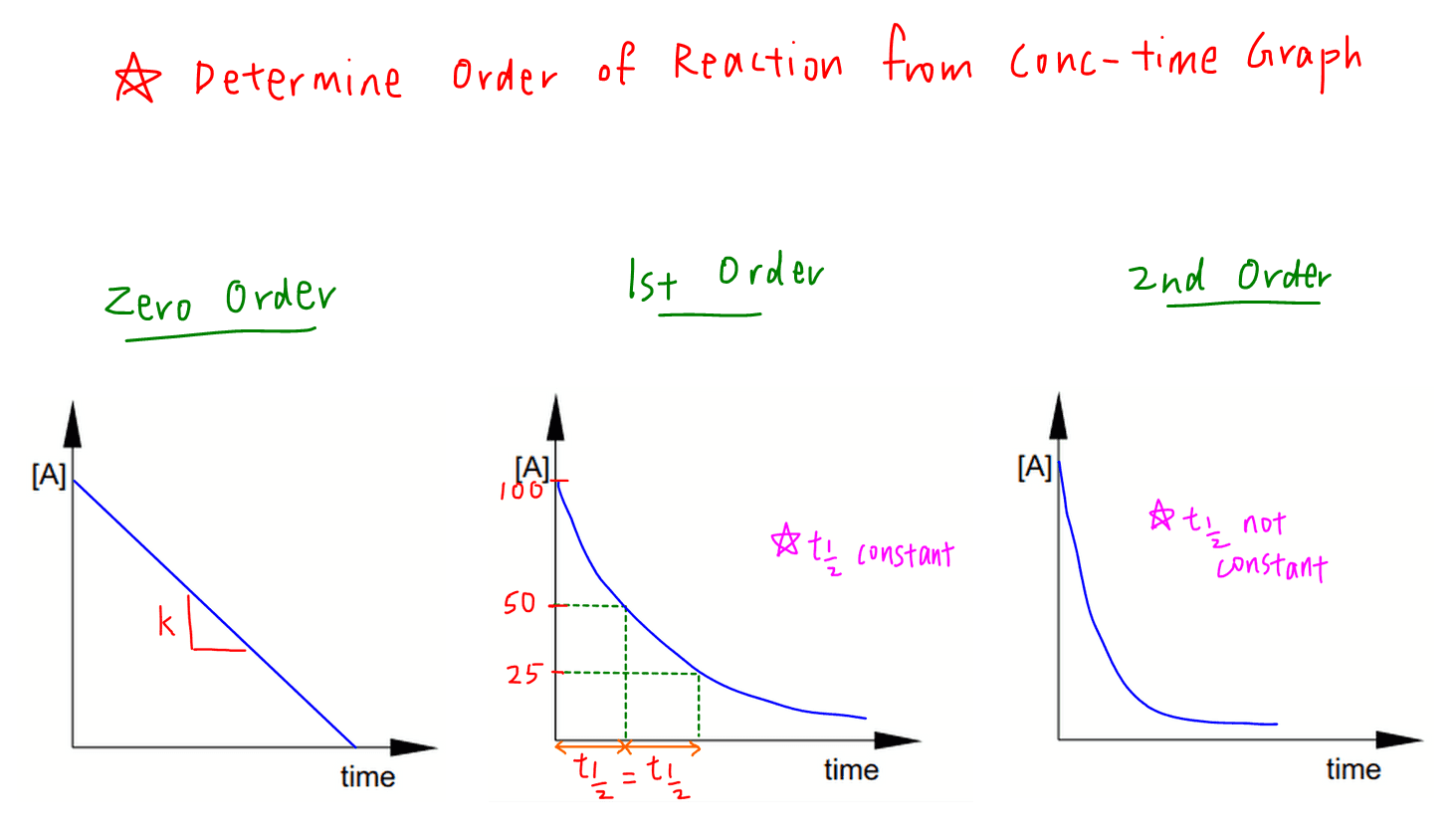
N t N 0 05 t n. T1 2 0693 λ t 1 2 0693 λ. We can determine that the order of a decay reaction is 1 by looking at the.
For a zero order reaction A products rate k. This expression works best when the number of half-lives is a whole number. An archaeological artifact containing wood had only 80 of the 14C found in a living tree.
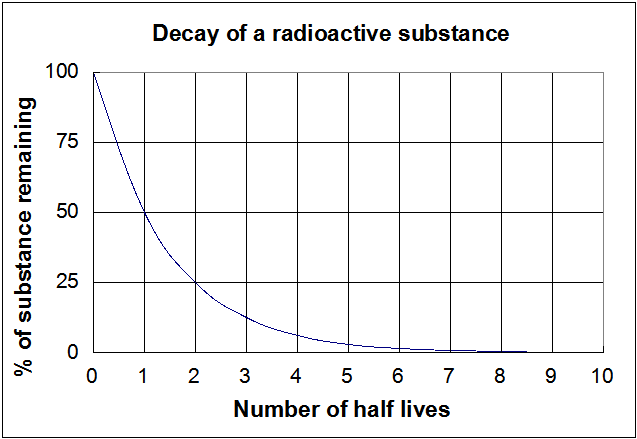
Half-life symbol t 12 is the time required for a quantity to reduce to half of its initial valueThe term is commonly used in nuclear physics to describe how quickly unstable atoms undergo. The formula for half-life in chemistry depends on the order of the reaction. Converting a Half Life to a Rate Constant.
So the value of t can be found or determined using half life equation. We know that at the half-life time eqt_12 eq the concentration of the reactant will. N t is the remaining quantity of a substance after time t has elapsed.
Given half life of the substance is t1 2 t 1 2 004. Each person flips a coin and. Using the concentration-time equation for a second-order reaction we can solve for half-life.
A second-order reaction is a reaction whose rate is dependent on either of two cases. N 0 is the. For example if the half-life of a 500 gram sample is 3 years then in 3.
The rate law is dependent on the squared concentration of one reactant or the rate law is dependent on the. Rearranging the equation used in example 2 we. The formula for half-life in chemistry depends on the.
We are required to find N o when we have N t and t. Find the value of the decay constant of a radioactive substance having a half-life of 004 seconds. Half life formula chemistry Saturday July 30 2022 Edit.
Department of Chemistry Boston University Boston MA 02215 Using coin flips to count people Everyone in of a group of 202 people is initially standing. T is the half-life. Estimate the age of the.
The equations are given above. The half-life of a first-order reaction does not depend upon the. Where t12 is the half.
In nuclear reactions this time period. Substitute this information into the equation for the half life of a reaction with this order and solve for t ½.

How To Calculate Half Life Of A Second Order Reaction Chemistry Study Com
Radioactivity Half Life Lessons Blendspace

Half Life Formula And Radioactive Decay Reactions Medium
Half Life Introductory Chemistry

Chemical Kinetics 1 Chemical Kinetics Plan 1 The Subject Of A Chemical Kinetics 2 Classification Of Chemical Reactions 3 Determination Methods Of Ppt Download
Half Life Introductory Chemistry

5 Ways To Calculate Half Life Wikihow

How To Calculate Half Life Chemistry Tutordale Com
Rate Equation And Order Of Reaction
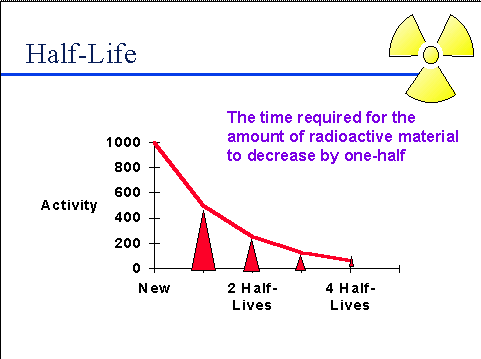
Radioactive Iodine 131 Has A Half Life Of Eight Days The Amount Of A 200 0 Gram Sample Left After 32 Days Would Be Socratic
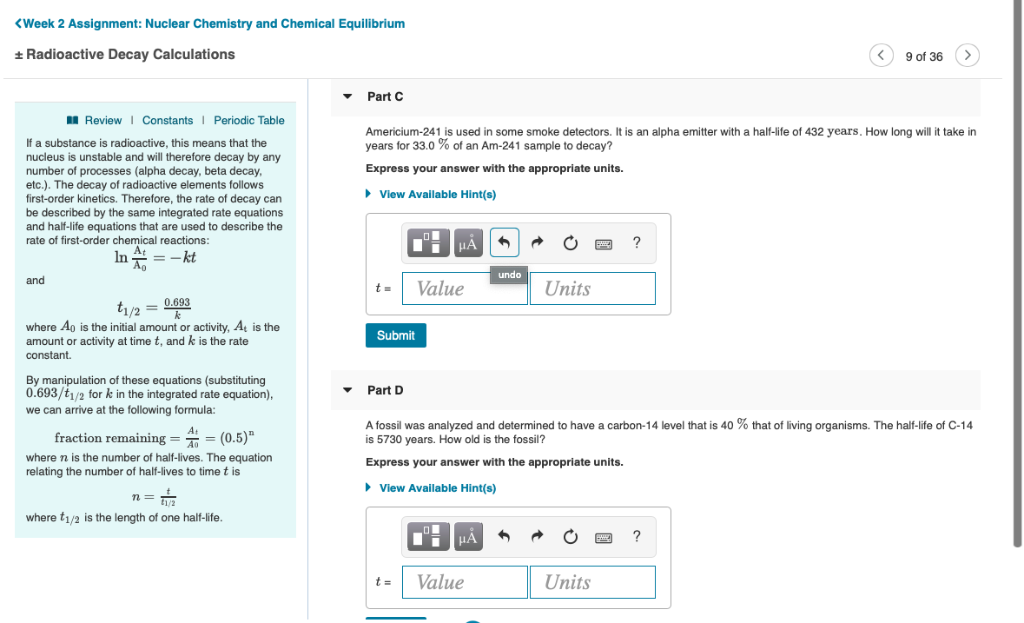
Solved Week 2 Assignment Nuclear Chemistry And Chemical Chegg Com

Radioactive Decay Model Math And Chemistry Science Activity Exploratorium Teacher Institute Project

How To Calculate Half Life Of A First Order Reaction Chemistry Study Com